On this slide I made two samples – one with cover-slip, and one left open to the air.
An open sample tends to be thicker and dries out pretty quickly – making it difficult to assess individual cell condition, and also allows the possibility of an airborne contaminant to confuse you.
Here’s a local fiber filament resting on top of a cover-slip. It was an important discovery that alerted me to the contamination possibility and prompted me to do a sticky-tape sampling of my vicinity. There were plenty of contaminant possibilities. The clue here was the shorter focal length above the actual sample i.e. on top of slide, (though the hyrdogel filaments are fairly discernible by there structure)
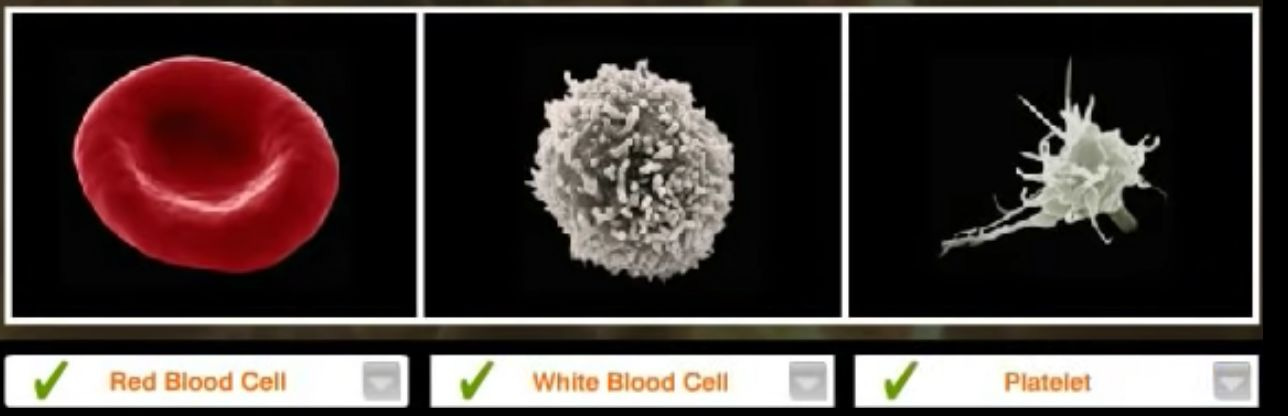
Under a cover-slipped blood sample there is generally only a few things you should see -
Red blood cells (corpuscles) – Erythrocytes - a normal red cell is approximately six to eight microns in diameter
White blood corpuscles – Leucocytes > are bigger than red cells. The granulocyte category are the neutrophils, the eosinophils and the basophils, because their cytoplasm has granules.
Lymphocytes and monocytes are mononuclear cells because generally they don't present with granules in the cytoplasm.
Blood platelets – thrombocytes - platelets are usually about a quarter or a fifth the diameter of a normal red cell. Some are Large platelets, and a Giant platelet is bigger than a normal red cell.
Other possibilities are:
chylomicrons – fat particles – tiny specks adrift in plasma.
Spicules - fine lines in clumps in plasma – sign of ill health
Rouleaux – stacked red blood cells
mis-shapen blood cells – oval, crinkled.
And of course the Plasma – which looks straw coloured, or clear under the microscope.
So when other ‘things’ turn up . . it gets (more) interesting.
This was the covered sample:
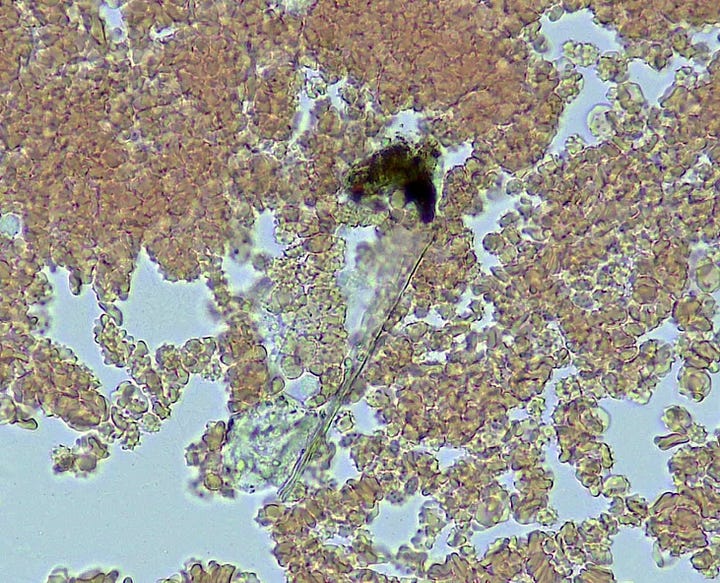
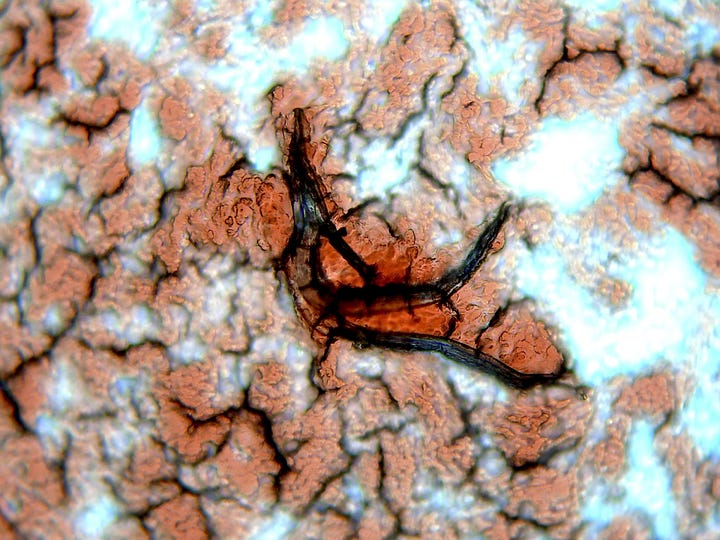

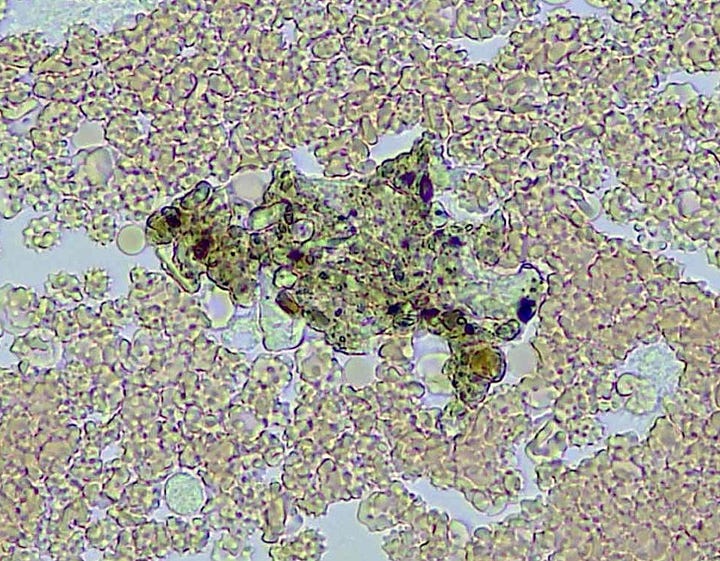
Note the White blood cells tackling ‘whatever this is’.
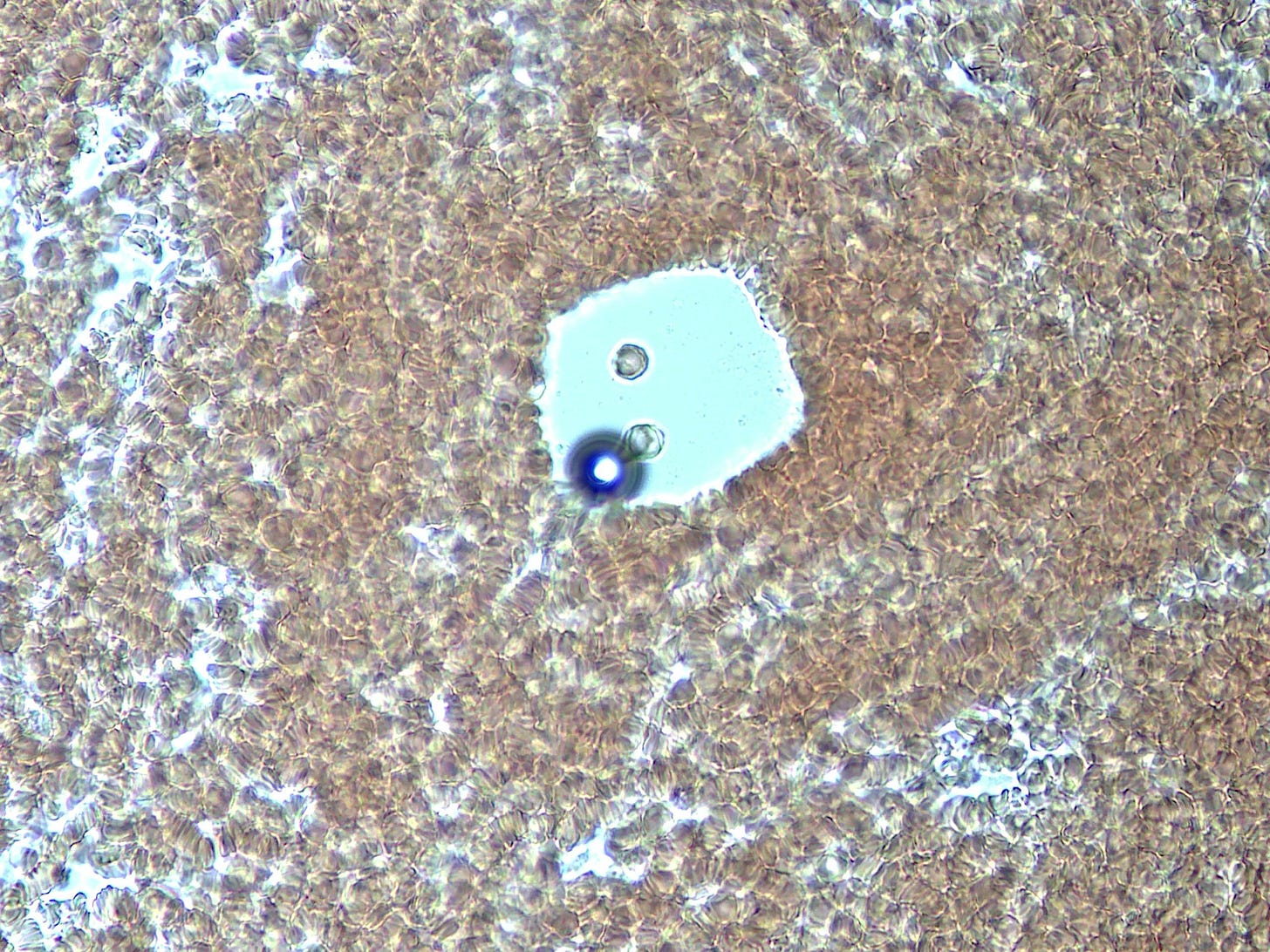
And then there is the spontaneous appearance of these ‘bubble islands’ (hydrogel islands?). I would be focused on a small area when suddenly the cells and plasma would start to flow across the field of view (FOV). A zoom out and scan around would reveal that these bubbles had begun forming, and displacing the fluid under the cover-slip.
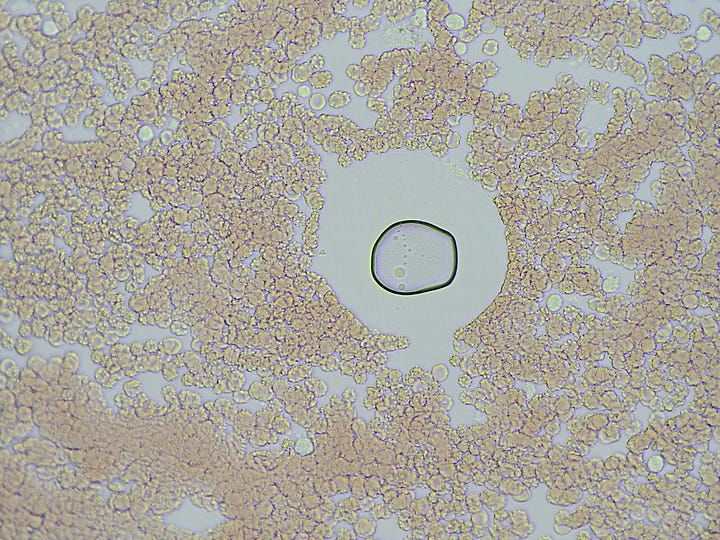
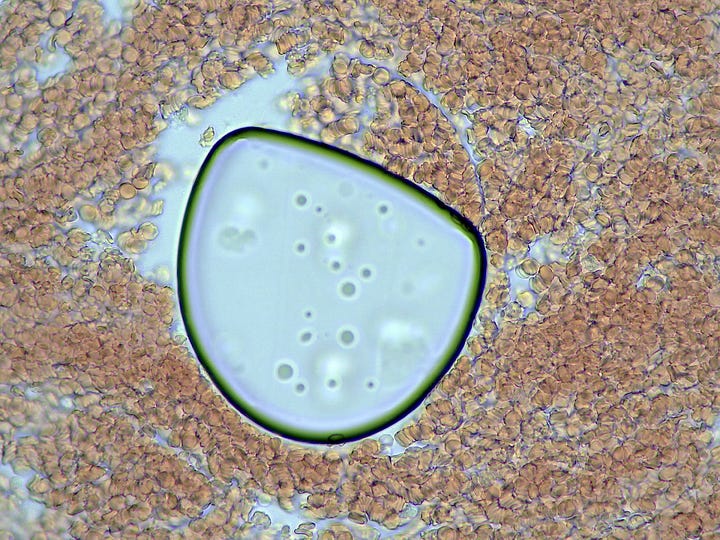
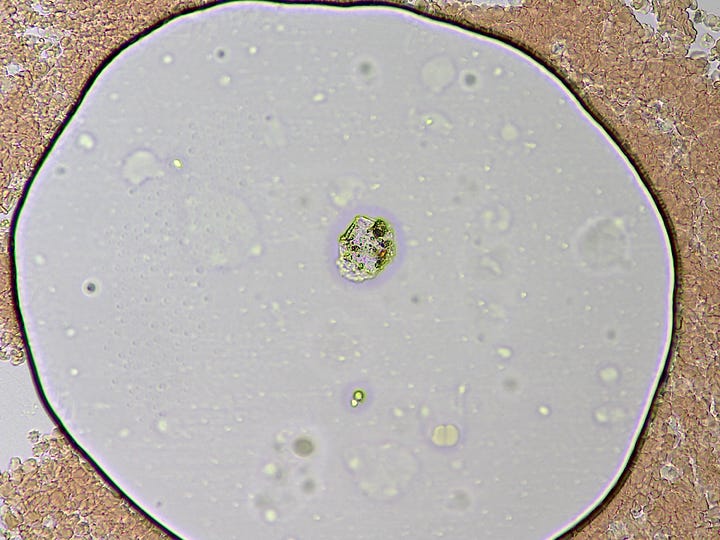
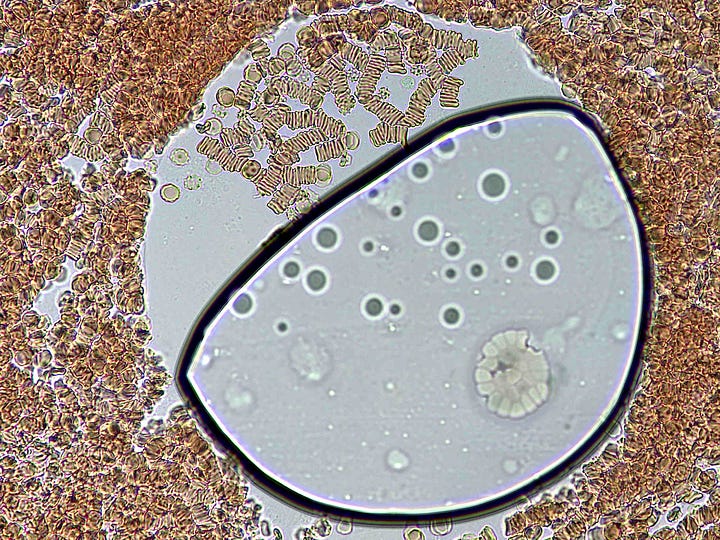
How they form and why was a mystery – they are not air bubbles. They are fluid filled and have a bounding membrane. Some open like a gate to allow cells to enter, then close again. Others stay closed. Some reform for no apparent reason.
This particular ‘bubble island’ (below) developed this refracted light spot, and began interacting with what looks like a distended white blood cell. Over the course of my obzervation it allowed more cells to enter.
I touched the cover-slip in the first video which demonstrates the integrity of the boundary.
The 2nd video shows some cell inclusions and the beginning of ‘tiny things’ getting active within.
Huh ! Then all of sudden the ‘light spot’ began to diminish over about 5 seconds and disappeared !
The 3rd video is a revisit 45 hours later – showing a dramatic increase in the number of very active ‘tiny things’ !!
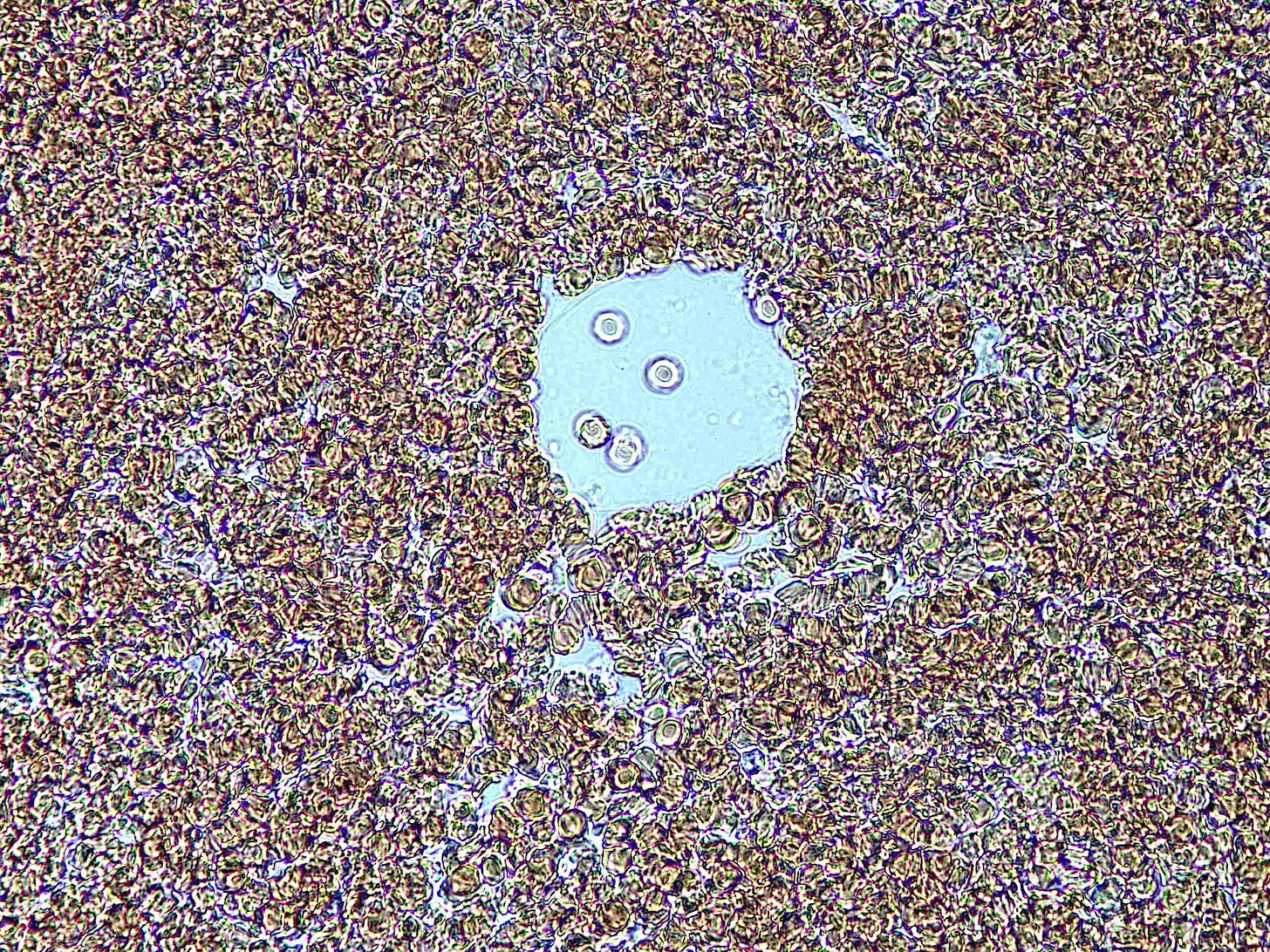
Below: Img. 1 - this bubble island seems to have replaced a previous one. Note the heavily rouleauxed RBCs surrounding it. Img. 2 & 3 – Unusual cellular colonies growing over time within the bubbles.
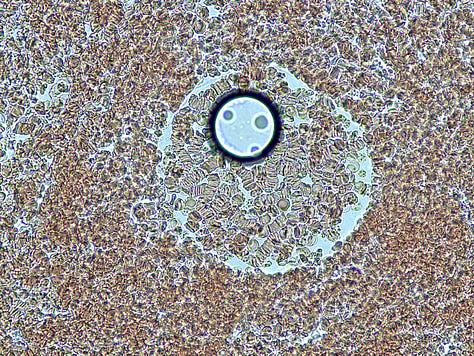
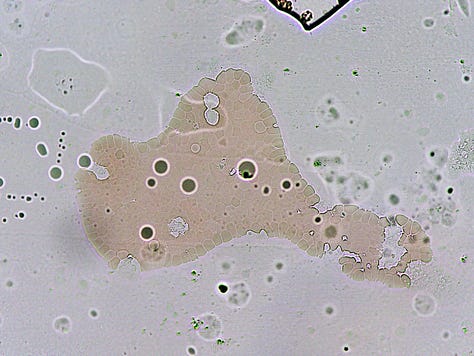
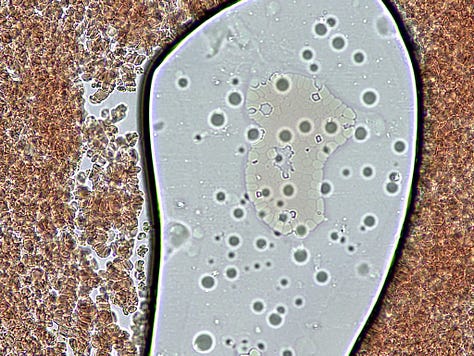
A standard cover-slip is 18 mm x 18 mm – about the area of a 5c piece. At 400x magnification the FOV (field of view) of my microscope is about 0.5 of a mm (500 μm). That’s 36 FOVs x 36 FOVs (1,296 separate FOVs) to fully scan one cover-slipped sample at that magnification !!
Fortunately I can scan at 40x (5mm FOV) and identify broader areas of interest initially.
I’d like to offer a relatable perspective of magnification – the tiny speck on my little finger-nail is the same Mullein seed at 400x.


Next time I’ll look at some various body fluids . . .



Hi MZ,
you can buy a darkfield patch kit for 30 odd $ and see more.
Best regards, matt.
Very interesting pictures! Do you have a video of this? :'Some open like a gate to allow cells to enter, then close again.' I've never seen that before.
In the hydrogels pictures where there are 4 pictures: is the hydrogel in the bottom right corner maybe shrunk? I've only seen that type of hydrogel once in my own samples, when it dilated from the heat of the lens, then shrunk back. Otherwise all other hydrogels spread, never withdrawn. Maybe this gel in the picture is like that?